| Photocatalytic conversion of carbon dioxide into methanol using optimized layered double hydroxide catalyst |
Naveed Ahmed, Motoharu Morikawa, and Yasuo Izumi,
Catalysis Today, 185(1), 263– 269 (2012) ![]() [The
PDF file]
[The
PDF file]
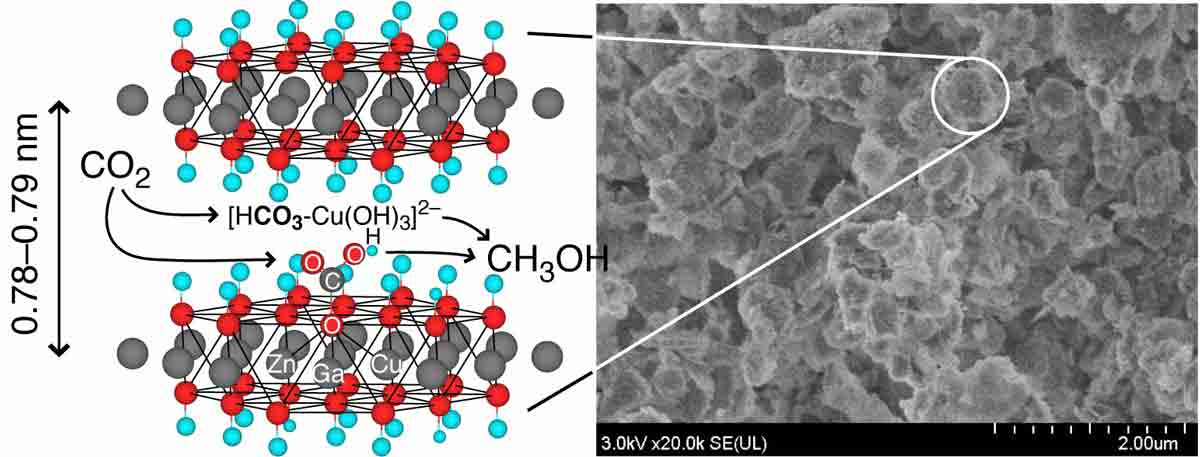
The photocatalytic reduction of carbon dioxide into methanol was enabled between the Zn–Ga or Zn–Cu–Ga hydroxide layers using hydrogen and was promoted by the partial desorption of structural water stuffed between the cationic layers. The photoreduction rate obtained using [Zn1.5Cu1.5Ga(OH)8]+2(CO3)2–·mH2O was improved by replacing interlayer carbonate anions with [Cu(OH)4]2– to 0.49 mmolMethanol h–1 gcat–1, and the methanol selectivity was 88 mol%. At the molar level, interlayer Cu species was 5.9 times more active than the octahedral Cu sites in the cationic layers. The bandgap value was evaluated as 3.0 eV for the semiconductor [Zn1.5Cu1.5Ga(OH)8]+2[Cu(OH)4]2–·mH2O. Direct electronic transition from O 2p to metal 3d, 4s, or 4p was responsible for the photocatalysis excited largely by ultraviolet (UV), and to a lesser extent by visible light.
Chiba University > Graduate School of Science > Department of Chemistry > Dr. Yasuo Izumi Group